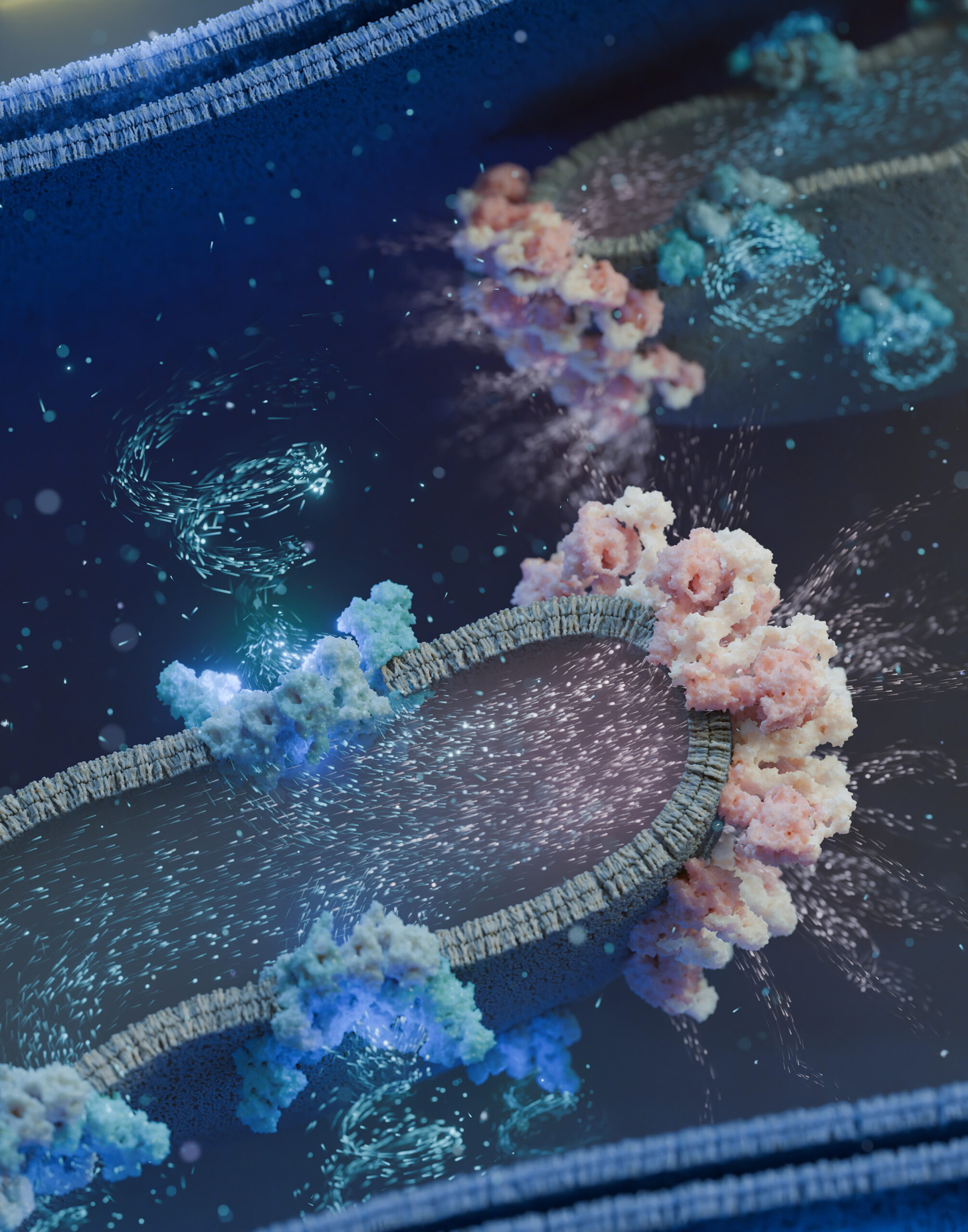
Mitochondria are the power stations in our cells. They produce the energy for all vital processes. Using cryo-electron tomography, researchers at the University of Basel have now gained insights into the architecture of mitochondria with unprecedented resolution. They discovered that the proteins responsible for energy production assemble into large ‘supercomplexes’. These supply the cell with the necessary energy.
Basel/Switzerland, March 21, 2025 Most living organisms on our planet – whether plants, animals or humans – have mitochondria in their cells. These provide the energy for almost all cellular processes. To do this, they use the oxygen we breathe and carbohydrates from food to produce ATP, the cells‘ universal energy source. Strictly speaking, this task is performed by proteins that work together in the so-called respiratory chain.
Although the respiratory chain was discovered 70 years ago, its exact organisation within the mitochondria was previously unclear. Using state-of-the-art cryo-electron tomography, researchers led by Dr Florent Waltz and Prof. Dr Ben Engel at the Biozentrum of the University of Basel have now succeeded in capturing high-resolution images of the respiratory chain directly in cells at an unprecedented resolution. The results of the study have been published in ‘Science’.
Architecture ensures efficient energy production
‘Our data show that the proteins of the respiratory chain organise themselves in certain membrane regions of the mitochondria, stick together and form supercomplexes,’ explains Florent Waltz, SNSF Ambizione Fellow and first author of the study. ‘Some of these supercomplexes were clearly visible under the electron microscope – we were able to recognise their structures directly and elucidate their function. These supercomplexes pump protons through the mitochondrial membrane. The ATP-producing proteins, which work like a water mill, ultimately utilise this proton flow to produce ATP.’
The researchers analysed mitochondria in living cells of the algae Chlamydomonas reinhardtii. ‘We were very surprised that all proteins are actually organised in such supercomplexes,’ says Waltz. ‘This architecture could make ATP production more efficient, optimise electron flow and minimise energy loss.’
In addition to the supercomplexes, the researchers were also able to analyse the membrane architecture of the mitochondria in more detail. ‘It is somewhat reminiscent of lung tissue: the inner mitochondrial membranes are highly folded, which increases the surface area to accommodate as many respiratory chain proteins as possible,’ says Ben Engel.
New perspectives for evolution and health
In future, the researchers want to find out why the supercomplexes in the respiratory chain are connected to each other and how this synergy increases the efficiency of cellular respiration and energy production.
The study could also provide new insights for biotechnology and diseases. ‘By studying the architecture of these supercomplexes in other organisms, we can gain a more comprehensive understanding of their basic organisation,’ explains Waltz. ‘This could not only reveal evolutionary adaptations, but also help to understand how disruptions to this architecture play a role in certain diseases in humans.’
Original publication:
Florent Waltz, Ricardo D. Righetto, Ron Kelley, Xianjun Zhang, Martin Obr, Sagar Khavnekar, Abhay Kotecha, Benjamin D. Engel: In-cell architecture of the mitochondrial respiratory chain, Science, March 21, 2025. doi: (https://doi.org/10.1101/2024.09.03.610704)
ImageSource
Biozentrum / Verena Resh, (https://luminous-lab.com/), The proton flow from supercomplexes of the respiratory chain (blue) to ATP complexes (pink) enables ATP production in the mitochondria.